
(MONMOUTH JUNCTION, N.J.) – A New Jersey-based pharmaceutical company has expanded a November 2018 recall of lots of liquid infant ibuprofen that might be more concentrated than advertised.
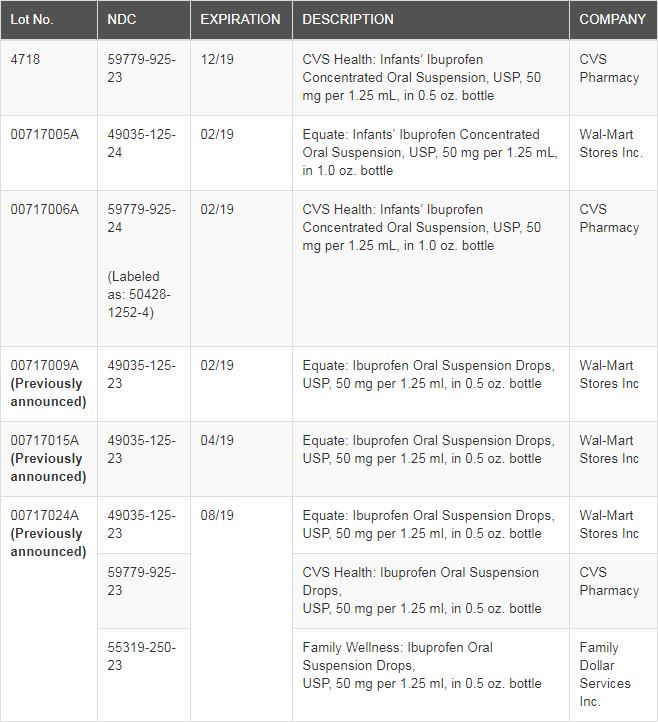
Tris Pharma’s voluntary recall covers Infants’ Ibuprofen Concentrated Oral Suspension, USP (NSAID) 50 mg per 1.25 ml. Three lots have been added to the original recall.
The company says there is a “remote possibility” that infants more susceptible to medication with a higher potency of ibuprofen might suffer permanent renal injury.
“Adverse effects that may be experienced are nausea, vomiting, epigastric pain, or more rarely, diarrhea,” according to Tris Pharma in November. “Tinnitus, headache and gastrointestinal bleeding are also possible adverse effects.”
The company says that to date, no serious adverse events have been reported in relation to this recall.
Consumers, who may be concerned, should contact their physician or healthcare provider if they have experienced any problems that may be related to taking or using this drug product.
Consumers with questions regarding the recall can contact Tris Customer Service at (732) 940-0358 (Monday through Friday, 8:00 a.m. ET- 5:00 p.m. PT) or via email at micc_tris@vigilarebp.com.



